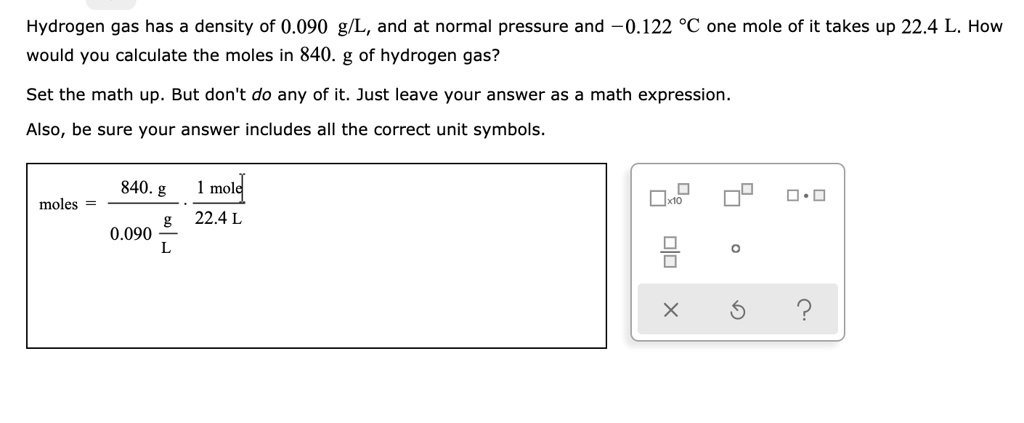
calculate number of moles of 10g of hydrogen and 64 g oxygen.also find limiting reagent when hydrogen and oxygen reacts to give water

21. The number of gram atoms of sulphur prese in 3 mole of hydrogen sulphide is begin{array}{lllll}text { 1) } 3 & text { 2) } 2 & text { 3) } 1.5 & text { 4) } 1.0end{array}

How many grams of hydrogen gas are needed to produce 5.00 g of water according to this equation? - YouTube

Atomic Mass and The Mole Topic: AMU's & Atomic Mass Objectives: Day 1 of 3 To learn how we define 1 amu (atomic mass unit) To learn how we derive atomic. - ppt download

5. Which of the following statements is incorrect? (1) one gram atom of carbon contain Avogadro's number of atoms. (2) one mole of oxygen gas contains Avogadro's number of molecules. (3) one